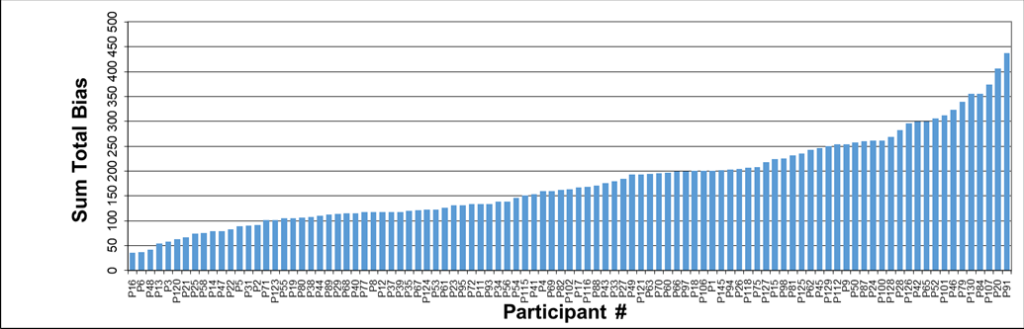
The Reynolds Cup (RC) competition continues to grow in popularity and of 120 sets distributed for the 12th RC, 99 contestants, spanning 30 different countries, returned results. Each set consisted of three, very carefully prepared, samples designed to simulate a kaolin, a bentonite, and a sandstone, all common clay mineral bearing sample types that pass regularly through many analytical labs. As in previous RC’s, the sum of the absolute total difference between the known concentrations of the mineral components of the samples and the concentrations submitted by each participant, plus any misidentified minerals, was used to calculate a ‘sum total bias’.
As usual, any method or combination of methods is allowed, though almost all participants rely mainly on X-ray powder diffraction. The top finishing labs, all with an impressive bias of < 60 were as follows:

5th place (bias 58.5) Youjun Deng (center) and his team from left to right – Camilo Jose Sanchez Avellaneda, Md Wasek Foysal, Chia-Wei Lin, Ayazhan Bazarbekova, from Texas A&M University.

4th place (bias 55.0) Kristian Ufer, Stephan Kaufold, and Reiner Dorhmann from the BGR, Hannover. From 3rd place owards, partcipants recieve an individual Reynolds Cup plaque and have their details recorded on a brass plaque on the Reynolds Cup box.

In 3rd place (bias 42.6), was Gi Young Jeong from Andong National University, Republic of Korea.

2nd place (bias 37.4) was Rieko Adriaens, Gilles Mertens, Silja Frederickx, Ines Boushah, Nathalie Lan Dung, Nancy Weynds, Ithar Shabeeb, Dania Alkhalidy, Abdullah Mohammad, Dominique Jacques, and Bram Paredis from Qmineral, in Belgium

And finally in 1st place, the winner of the 12th Reynolds Cup competition C (drum roll!), with a bias of 36.4, was Mark Raven and the team of Peter Self, Rodrigo Gomez-Camacho, Rachael Maree, Rong Fan, Nick Owen, and Shu Huang from CSIRO in Adelaide.
How did they do it?
The as received samples (Figure 1) were initially weighed to determine the amounts available for testing (Table 1).

Figure 1. As received RC12 samples.
Sample weights for testing
| Sample Description | As received (g) | Micronized (g) | Clay Separation (g) | Reserved (g) | Corundum added (wt%) | |
| RC12-1 | Kaolin | 4.5 | 1.5 | 2 | 1.0 | 25.05 |
| RC12-2 | Brown bentonite | 4.0 | 1.5 | 2 | 0.5 | 25.15 |
| RC12-3 | Old red sandstone | 4.2 | 1.5 | 2 | 0.7 | 25.02 |
Preliminary XRD testing was performed by lightly back pressing the ‘as received’ samples into stainless steel holders for analysis. This was done to ascertain pristine mineral components before further preparation procedures. XRD patterns were collected on a PANalytical X’Pert Pro MPD using iron filtered cobalt K alpha radiation in both auto and fixed divergence slit modes. Patterns were collected from 4 to 80° 2‑theta at 0.017° per step. Semi-quantitative elemental analysis using a Spectro energy dispersive XRF was also initially performed under helium on loose powder ‘as received’ samples to help confirm minor phases that may otherwise be difficult to identify by XRD alone. Additional multi-element analysis of the reserved ‘as received’ sub-samples were performed using lithium borate fused beads on a Bruker Tiger wavelength dispersive XRF.
Sub-samples of 1.5 g of each of the samples were micronized for 10 minutes under ethanol with a McCrone micronizing mill then oven dried at 60° C. After drying, the micronized samples were thoroughly mixed in an agate mortar and pestle to ensure homogeneity. The fine powders were lightly back pressed to minimise preferred. The process of micronizing the samples under ethanol followed by oven drying partially dehydrates most swelling clay minerals resulting in broad asymmetric 00l peaks. The micronized samples were subsequently calcium saturated to restore the 001 peaks of the main swelling clay minerals to ~15 Å. This was achieved by washing the micronized samples twice with 1M CaCl2, washing with deionised water followed by ethanol (centrifuged at 6000 rpm after each step) before oven drying at 60° C. The Ca saturated samples were again thoroughly mixed in an agate mortar and pestle to ensure homogeneity and lightly back pressed into sample holders for XRD measurement. XRD patterns were then re-collected. Comparison of the XRD patterns before and after Ca saturation also helped identify water soluble phases (i.e. halite) in sample RC12-3.
Additional 2 g sub-samples of the as received material were dispersed with 1M NaCl and repeatedly centrifuged at different speeds to separate various size fractions. Samples RC12-1 and RC12-2 were separated into the following fractions: <0.2 µm, 0.2-2 µm and >2 µm. Initial QXRD of sample RC12-3 indicated low total clay content so only the <2 µm and >2 µm fractions were separated. The fractions were Ca saturated and prepared as pressed powder, oriented, calcium and magnesium saturated and glycerolated to help identify the clay mineral species. Oriented Mg and glycerolated XRD patterns of the <0.2 µm fraction of RC12-3 indicated interstratified species which was modelled in NEWMOD2 with regularly interstratified illite-smectite clay. Halloysite was primarily identified in the 0.2-2 µm and <0.2 µm fractions of sample RC12-1 by a broad shoulder on the low angle edge of the kaolinite peak at ~7.3 Å. This was later confirmed using formamide expansion on an oriented specimen of the <0.2 µm fraction of RC12-1. Dioctahedral smectite (montmorillonite) was confirmed in powder samples of the <0.2 µm fractions of RC12-1 and RC12-2 using the position of 060 peaks at ~1.499 Å. A broad shoulder on the low angle edge of the montmorillonite peak in RC12-2 was overlooked as nontronite. Quantification was performed on all RC sub-samples and size fractions using TOPAS version 7. Disordered clay minerals were calibrated from purified standards using PONKCS (Partial Or No Known Crystal Structures) procedures. Amorphous content was determined using the internal standard method after the addition of ~25 % by weight (Table 1) of a fine-grained corundum standard to each micronized Ca saturated sample. Values of amorphous content less than 5% were ignored in the final composition of RC12-3.
